Equitable Care for Patients
Equitable Carrier Screening with NxGen MDx
NxGen MDx’s carrier screening is focused on providing equitable care, regardless of a patient’s ethnicity or family history. Taking into consideration evidence-based guidelines from multiple societal bodies, NxGen MDx has curated panels designed to meet the needs of a multi-ethnic population, provide actionable results, and improve patient outcomes.
Early Advantage Panel
The Early Advantage Panel from NxGen MDx is an innovative approach to carrier screening panel design because it is the first of its size to incorporate conditions that the U.S. Department of Health and Human Services (HHS) recommends be included in newborn screening (NBS), reiterating our focus on improving pregnancy and newborn health outcomes for families everywhere.
The list of conditions that the HHS recommends all states use in their NBS programs is known as the Recommended Uniform Screening Panel (RUSP) and was compiled based on evidence that supports the benefits of early diagnosis and the availability of effective treatments.
- Screens for 68 conditions, including those found on the RUSP.
Super Panel
The Super Panel prioritizes intelligently curated gene selection and comprehensive technology to provide an equitable carrier screening panel for all patients, regardless of their ethnicity or family history.
- Screens for 145 conditions that are common, severe, early onset, and are clinically actionable.
- At-risk couple detection rate of 1 in 23, one of the highest in the industry.
Find out more about NxGen MDx’s carrier screening panels!
Carrier Screening is For All Patients
The American College of Obstetricians and Gynecologists recommends that
“Information about genetic carrier screening should be provided to every pregnant woman.”1
The American College of Medical Genetics and Genomics states that “Carrier screening paradigms should be ethnic and population neutral and more inclusive of diverse populations to promote equity and inclusion.”2
| Condition(s) | Incidence (# of Pregnancies) |
|---|---|
| Recessive and X-Linked Conditions | 1 in 3003 |
| Down Syndrome | 1 in 800 |
| Neural Tube Defects | 1 in 1000 |
| Cystic Fibrosis | 1 in 3500 |
Strengthening Newborn Screening (NBS)
NBS is one of the most successful public health initiatives in the United States.
It is the nation’s largest genetic screening program and screens approximately 4 million infants every year. While NBS is an important part of neonatal care, it does have some limitations.
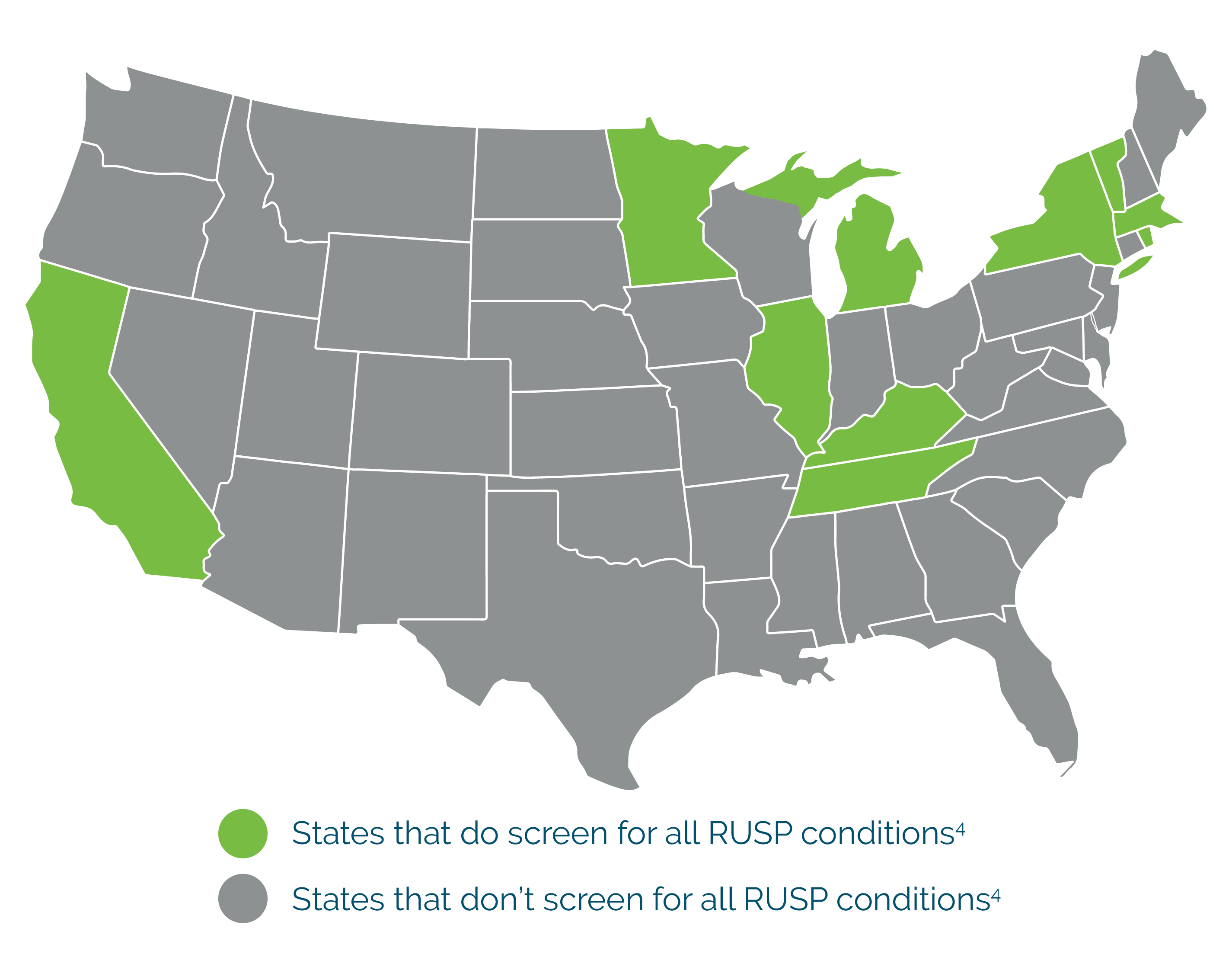
- The number of conditions screened for is inconsistent from state to state.
- NBS can be negatively impacted by several different factors such as birth weight, ethnicity, and time of sample collection that can lead to more false positives & false negatives.
- Results may be returned too late to begin treatments that are critical in the first few days of life.
As a healthcare provider, when you know a child’s risk for a genetic condition before they are born, you can work with parents to come up with a birth and early care plan supplemented by targeted diagnostic testing.
NBS in the United States

Using Carrier Screening (CS) to Address Gaps in NBS
| Benefits | NBS | CS |
|---|---|---|
| Identifies At-Risk Children Early in Life | ✓ | ✓ |
| Provides More Reproductive Options for Parents | ✓ | |
| Provides More Diagnosis Options During Pregnancy or at Delivery | ✓ | |
| Allows for Birth and Delivery Options | ✓ | |
| Treatment Immediately After Birth | ✓ | |
| Lower False Negatives | ✓ | |
| Lower False Positives | ✓ | |
| Health Equity Regardless of State of Residence | ✓ |
NxGen MDx Carrier Screening in Your Practice
If you are interested in offering NxGen MDx carrier screening in your practice, or want to know more about the company click below to contact a NxGen MDx representative.
Sources
1- Committee Opinion No. 691: Carrier Screening for Genetic Conditions. Obstet Gynecol. 2017 Mar;129(3):e41–55.
2- Gregg AR, Aarabi M, Klugman S, Leach NT, Bashford MT, Goldwaser T, et al. Screening for autosomal recessive and X-linked conditions during pregnancy and preconception: a practice resource of the American College of Medical Genetics and Genomics (ACMG). Genet Med. 2021 Oct;23(10):1793–806
3- Johansen Taber KA, Beauchamp KA, Lazarin GA, Muzzey D, Arjunan A, Goldberg JD. Clinical utility of expanded carrier screening: results-guided actionability and outcomes. Genet Med. 2019 May;21(5):1041–8.
4- “Newborn Screening.” NORD (National Organization for Rare Disorders). 26 Jan. 2021. https://rarediseases.org/policy-issues/newborn-screening/
Accurate Screening for All Pregnancies
- NIPS can be used in singleton and twin pregnancies
- High sensitivity and specificity regardless of age, ethnicity, or BMI
- No restrictions for ART pregnancies, egg donors, surrogates, or bone marrow/organ transplant recipients
- Low “no-call” (assay failure) rate (0.3%) means fewer unnecessary diagnostic procedures
%
No-Call Rate
Common Trisomies & Sex Chromosomes (Standard)
The Informed Prenatal Screen looks at these conditions
- Trisomy 21 (Down Syndrome)
- Trisomy 18 (Edwards Syndrome)
- Trisomy 13 (Patau Syndrome)
- Fetal sex (including individual fetal sex calling for twin pregnancies*)
- Monosomy X (Turner Syndrome)
- Trisomy X (Triple X Syndrome)
- XXY Syndrome (Klinefelter Syndrome)
- XYY Syndrome (Jacob’s Syndrome)
Further Screening Options (Upon Request)
Expanded Aneuploidy Analysis
Expanded aneuploidy analysis may offer insight into pregnancies with fetal growth restriction or unexplained ultrasound abnormalities.
Microdeletions
- 22q11.2 (DiGeorge Syndrome)
- 15q11.2 (Angelman/Prader-Willi Syndrome)
- 1p36 Deletion Syndrome
- 4p (Wolf-Hirschhorn Syndrome)
- 5p (Cri-du-chat Syndrome)
Superior Platform, Superior Screen
Prenatal Screening with WGS (whole genome sequencing) has a significantly lower no-call rate than prenatal screening using a single nucleotide polymorphism (SNP) based methodology. Having a low no-call rate reduces the number of invasive procedures that are needed and decreases your patients’ anxiety.
Features of WGS vs. SNP Methodology
| Features | SNP | WGS |
|---|---|---|
| Common Chromosomal Aneuploidies | ✓ | ✓ |
| Sex Chromosome Aneuploidies | ✓ | ✓ |
| Fetal Sex | ✓ | ✓ |
| Microdeletions | ✓ | ✓ |
| Expanded Aneuploidy (23 Chromosomes) Analysis | ✓ | |
| No Limitations Based on BMI | ✓ | |
| Most Widely Validated by Leading Medical Institutions | ✓ | |
| Fewer Unnecessary Invasive Procedures | ✓ |


Actionable Results for All Your Patients
We use a whole genome sequencing (WGS) platform because we believe that all patients deserve an NIPS result that they can rely on.


Individual Fetal Sex Determination for Twins*
NxGen MDx can determine the sex of both fetuses in the case of a twin pregnancy.*
%
High 22q11.2 Positive Predictive Value
NxGen MDx Informed Prenatal Screen’s positive predictive value (PPV) is 90.0% for 22q11.2 deletion syndrome.
Connect Your Patients with a Certified Genetic Counselor
When you work with us, you and your patients will have access to a team of board-certified genetic counselors. Our counselors are available to discuss the results of screens and what they mean. Patients can discuss their results by scheduling an appointment with one of our counselors.
Connect With Us
If you are interested in offering the NxGen MDx Non-invasive Prenatal Screen or want to learn more, click the button below to contact a NxGen MDx representative.
Sources
*Presence/absence of chrY is detected at >99.9% accuracy. Fetal sex discrimination between, Male/Male, Male/Female, and Female/Female has an overall accuracy of 91%. -NxGen MDx Internal Data 2022.
Clear and Actionable Inherited Cancer Risk Assessment
Some cancers are hereditary, meaning that your patients can inherit genetic variants which can increase the risk of developing certain cancers in their lifetime.
Patients with a strong family history of cancer should be screened. Some indications for a strong family history of cancer include:
- Cancer at an early age.
- The presence of rare cancers such as ovarian or male breast cancer.
- Multiple relatives with the same type of cancer.
- One relative diagnosed with multiple primary cancers.
Screen & Evaluate
Paper & digital tools are available to identify patients who meet testing criteria. 1 in 4 patients who screen are likely to meet criteria.
Diagnose
NxGen MDx’s Hereditary Cancer Panel screens for 32 genes recommended by the National Comprehensive Cancer Network (NCCN).
Manage
Clinical management support includes pre- and post-test genetic counseling and reports detailing current NCCN management recommendations.
NxGen MDx Hereditary Cancer Testing in Your Practice
If you are interested in offering NxGen MDx hereditary cancer testing in your practice or want to know more about us, click below to contact a NxGen MDx representative.
NxGen MDx Tests for 99% of Community-Acquired Infections
NxGen MDx’s Vaginosis Test takes the guesswork out of diagnosis by providing you with a comprehensive picture of your patient’s vaginal microbiome and infection.
An internal review of more than 9,000 vaginitis samples showed that 70% of positive cases were coinfections.* By comparison, less thorough testing would have failed to identify these complex cases, potentially leading to an ineffective treatment plan and recurrence.
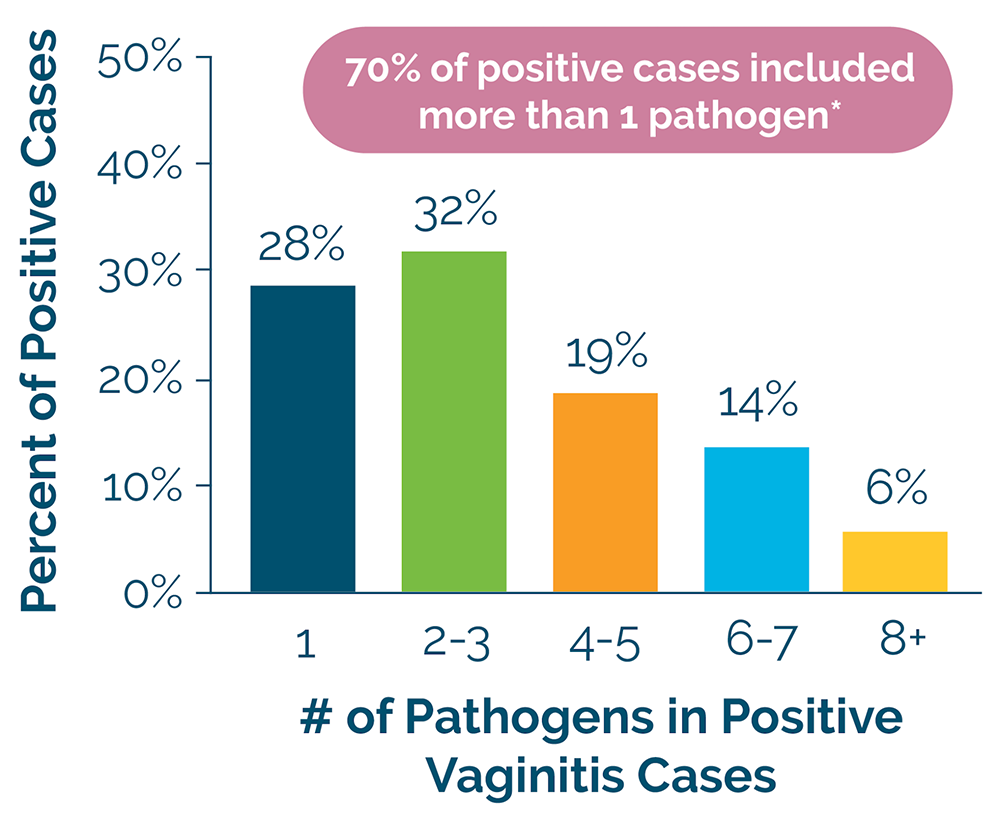

Use NxGen MDx’s Vaginosis Test and give your patient the optimal treatment plan for their unique infection the first time.
NxGen MDx Performs Simultaneous Antibiotic Resistance Testing


66% of more than 9,000 cases showed evidence of antibiotic resistance*
According to the CDC…
“Each year in the U.S., at least 2.8 million people get an antibiotic-resistant infection…”1
Stop the revolving door of patients coming in again and again with recurrent vaginitis. NxGen empowers you to practice antibiotic stewardship by testing for resistance to 10 classes of antibiotics and getting you a result in 24 hours.
Use NxGen MDx’s Vaginosis Test to have confidence in a treatment plan that is appropriate for each infection.
Included in NxGen MDx’s Vaginosis Test
Pathogens and Antibiotic Resistance Tested
Normal Flora
- Lactobacillus crispatus
- Lactobacillus gasseri
- Lactobacillus iners
- Lactobacillus jensenii
Candida (Yeast Infection)
- Candida albicans
- Candida dubliniensis
- Candida glabrata
- Candida krusei
- Candida lusitaniae
- Candida parapsilosis
- Candida tropicalis
Sexually Transmitted Infection
- Chlamydia trachomatis
- Mycoplasma genitalium ^
- Neisseria gonorrhoeae
- Treponema pallidum (Syphilis)
- Haemophilus ducreyi
- Herpes Simplex Virus 1
- Herpes Simplex Virus 2
- Trichomonas vaginalis
Pathogens Associated with Vaginosis
- Atopobium vaginae
- Bacteroids fragilis
- Enterococcus faecalis
- Escherichia coli
- Gardnerella vaginalis
- Megasphaera Type 1
- Megasphaera Type 2
- Mobiluncus curtisii
- Mobiluncus mulieris
- Mycoplasma hominis
- Ureaplasma urealyticum
- Prevotella bivia
- Staphylococcus aureus
- Streptococcus agalactiae (Group B Strep)*
- Bacteria Vaginosis-Associated Bacterium 2 (BVAB2)
Antibiotic Resistance
- Betalactam/Beta-lactamase
- Inhibitor Combination Drugs
- Carbapenems
- Extended Spectrum Beta-lactams
- Macrolides
- Methicillin
- Nitroimidazoles
- Penicillins & Cephalosporins
- Quinolone/Fluoroquinolone
- Sulfamethoxazole & Trimethoprim (SMX/TMP)
- Vancomycin
*The NxGen MDx Group B Strep Test can be ordered as a stand alone test, using the same technology and providing results with the same accuracy.
^This pathogen will still be tested for even when the patient opts out of STIs.
Bolded pathogens are included in the dirty catch urine collection option.
Reflex fluconazole sensitivity testing is performed for samples positice for C. albicans, C. dubliniensis, C. lusitaniae, C. parapsilosis, and C. tropicalis. As most C. glabrata and C. krusei isolates demonstrate fluconazole resistance, alternative treatment options are provided.
Vaginitis in Pregnant & Pediatric Patients
Untreated Vaginal Infections Impact Reproductive Health2,3
- Infertility
- Endometriosis
- Increased risk of acquiring STIs (including HIV)
- Preterm birth
- Neonatal sepsis
- Low birth weight
- Pelvic Inflammatory Disease
- Ventilation/respiratory distress in infants
Vaginitis is Common in Pediatric and Adolescent Patients4
50% of new STIs are in individuals under the age of 255. NxGen MDx offers a dirty catch urine collection option for pediatric patients and individuals for whom a vaginal wall swab is not an option.
- 10 pathogens
- Sexually Transmitted Infections (STIs)
- Yeast Infections
- Antibiotic Resistance (ABR)
The NxGen MDx Vaginosis Test Impacts Patient Care
The additional assistance with antibiotic resistance testing has also been beneficial as we have been able to treat appropriately to ensure patients will not have to repeat the evaluation as well as not having to take unnecessary medications.
The ability to evaluate normal flora has been so valuable and helps give us better direction when developing treatment plans.
Excellent customer service and any question or concern is always addressed in less than twenty-four hours. Thank you NxGen MDx!
With this comprehensive knowledge and quick turnaround time, I am able to treat my patients quickly and effectively, which creates an atmosphere of trust and reliability.
For years I felt limited to the testing that was available; however, the NxGen MDx swab testing evaluated my patient as a whole and ultimately limits the number of times my patients needs to come back for further testing and evaluation. We are able to treat the patient more effectively and quickly using this swab.
I have been using the NxGen MDx testing swab to diagnose vaginitis for the past 2-3 years. I have enjoyed the thoroughness of the swab that it will test for aerobic and anaerobic microorganisms at the same time as well as STI organisms.
The best part is the vaginosis panel includes treatment options and a resistance table to eliminate treatment failure.
The NxGen Vaginosis swab/panel allows me to get a complete analysis of the vaginal flora as well as any pathogenic organisms present. It also provides sensitivity for any pathogenic organisms, so patients are treated with the most effective course of action from the start.
The [NxGen MDx Vaginosis Test] is so easy to use and interpret, and the turnaround time is unbeatable. It has made such a difference for our patients who have struggled with recurrent infections to identify organisms that are not included in other swabs.
NxGen MDx Provides More than Just Results

Connect With Us
If you are interested in offering the NxGen MDx Vaginosis Test or simply want to learn more, click the button below to contact a NxGen representative.
Sources
*NxGen MDx. 2021. Data on file.
1- https://www.cdc.gov/drugresistance/index.html
2- Ravel J, Moreno I, Simón C. Bacterial vaginosis and its association with infertility, endometritis, and pelvic inflammatory disease. American Journal of Obstetrics & Gynecology. 2021 Mar 1;224(3):251–7.
3- STD Facts – STDs & Pregnancy Detailed Fact Sheet [Internet]. 2021 [cited 2021 Dec 20]. https://www.cdc.gov/std/pregnancy/stdfact-pregnancy-detailed.htm
4- Vulvovaginitis- presentation of more common problems in pediatric and adolescent gynecology (https://pubmed.ncbi.nlm.nih.gov/28927766/)
5- Sexually Transmitted Infections Prevalence, Incidence, and Cost Estimates in the United States (https://www.cdc.gov/std/statistics/prevalence-incidence-cost-2020.htm)
PCR Testing is More Reliable Than Culture
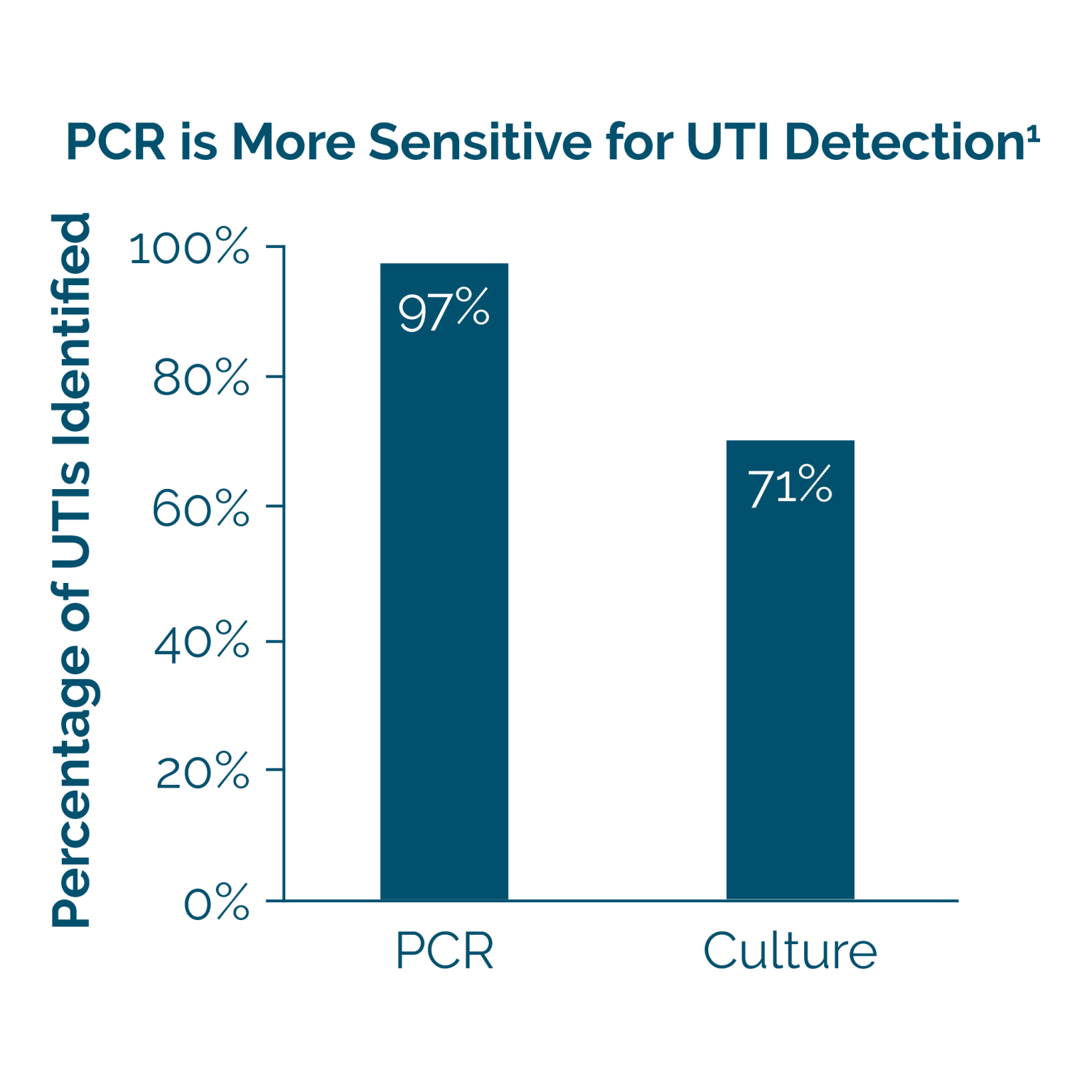

Using culture to diagnose a UTI is significantly limited by false-negative results and long turnaround times. NxGen MDx’s PCR-based UTI Test is about 25% more accurate than culture, giving you confidence in your patient’s results.1 Additionally, NxGen MDx’s PCR test includes simultaneous antibiotic resistance testing with a 24-hour turnaround time.
Use NxGen MDx’s UTI Test and give your patient the optimal treatment plan for their unique infection.
NxGen MDx Performs Simultaneous Antibiotic Resistance Testing
According to the CDC
“Each year in the U.S., at least 2.8 million people get an antibiotic resistant infection…”2
Stop the revolving door of patients coming in again and again with recurrent UTIs. NxGen MDx empowers you to practice antibiotic stewardship by testing for resistance to 10 classes of antibiotics with results in 24 hours.
Use NxGen MDx’s UTI Test to have confidence in a treatment plan that is appropriate for each infection.
%
of more than 1,200 cases showed evidence of antibiotic resistance³
Included in NxGen MDx’s UTI Test
Pathogens and Antibiotic Resistance Tested
Bacteriuria
- Acinetobacter baumannii
- Citrobacter freundii
- Enterobacter aerogenes
- Enterobacter cloacae
- Enterococcus faecalis
- Enterococcus faecium
- Escherichia coli
- Klebsiella oxytoca
- Klebsiella pneumoniae
- Morganella morganii
- Proteus mirabilis
- Proteus vulgaris
- Providencia stuartii
- Pseudomonas aeruginosa
- Staphylococcus saprophyticus
- Streptococcus agalactiae
Candiduria
- Candida albicans
Antibiotic Resistance
- Betalactam/Beta-lactamase Inhibitor Combination Drugs
- Carbapenems
- Extended Spectrum Beta-lactams
- Macrolides
- Methicillin
- Nitroimidazoles
- Penicillins & Cephalosporins
- Quinolone/Fluoroquinolone
- Sulfamethoxazole & Trimethoprim (SMX/TMP)
- Vancomycin
Reflex fluconazole sensitivity testing is performed for all samples positive for C. albicans.
Pregnancy Complications From UTIs
UTIs Can Impact Maternal and Fetal Health4-7
Pregnant women are at an increased risk for both symptomatic and asymptomatic UTIs. Therefore, all pregnant women should be tested and treated for bacteriuria to reduce the risk of serious complications which include:
-
- Pyelonephritis
- Hypertension/preeclampsia
- Anemia
- Chorioamniotis
- Low birth weight
- Premature labor
The NxGen MDx UTI Test Impacts Patient Care
Fosfomycin is included in the UTI sensitivity testing. In seven years of clinical practice, I have yet to see this offered elsewhere.
Partnering with NxGen MDx has positively impacted my clinical practice and patient outcomes.
NxGen MDx Provides More than Just Results


Connect With Us
If you are interested in offering the NxGen MDx UTI Test or want to learn more, click the button below to contact a NxGen MDx representative.
Sources
1 – Thermo Fischer Scientific. Urinary Tract Pathogen Detection: Reducing Time to Result [Internet]. [cited 2021 Dec 16]. https://www.fishersci.com/us/en/healthcare-products/webinars/urinary-tract-pathogen-detection-reducing-time-result.html
2 – CDC. Antibiotic Resistance Threatens Everyone [Internet]. Centers for Disease Control and Prevention. 2021 [cited 2021 Dec 16]. https://www.cdc.gov/drugresistance/index.html
3 – NxGen MDx. 2021. Data on file. n>1200
4 – Habak PJ, Griggs J. Urinary Tract Infection In Pregnancy. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 [cited 2021 Dec 21]. http://www.ncbi.nlm.nih.gov/books/NBK537047/
5 – Nicolle LE, Gupta K, Bradley SF, Colgan R, DeMuri GP, Drekonja D, et al. Clinical Practice Guideline for the Management of Asymptomatic Bacteriuria: 2019 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2019 May 2;68(10):e83–110.
6 – Moore A, Doull M, Grad R, Groulx S, Pottie K, Tonelli M, et al. Recommendations on Screening for Asymptomatic Bacteriuria in Pregnancy. CMAJ. 2018 Jul 9;190(27):E823–30.
7 – Szweda, H., & Jóźwik, M. Urinary Tract Infections During Pregnancy–An Updated Overview. Dev Period Med, (2016). 20(4), 263-272.






